|
3/16/2023 0 Comments Metals periodic table 
Because lithium is charged, it is now an ion and we write it like this: When lithium reacts, losing its outer shell electron, it now has 2 protons in the nucleus and 3 electrons, which means the overall charge is now +1. As protons have a positive charge and electrons have a negative charge, the overall charge is 0. Initially, lithium has 3 electrons and 3 protons. As the first shell is full at 2 electrons, lithium now has a full outer shell. When lithium reacts, it loses the electron in its outer shell, which means it has 2 electrons in the next outer shell. Lithium (a group 1 metal) has 3 electrons in total and 1 electron in its outer shell, making it highly reactive. Let’s look at some more examples Example – group 1 For example, let’s look at the metal sodium (Na) reacting with chlorine: This full outer shell will give them the same electronic structure as noble gases (group 0). When metals react, they lose electrons to gain a full outer shell. Transition metals (the middle section) – Less reactive metals.Most of the elements in the periodic table are metals, and we can broadly split them into two categories: 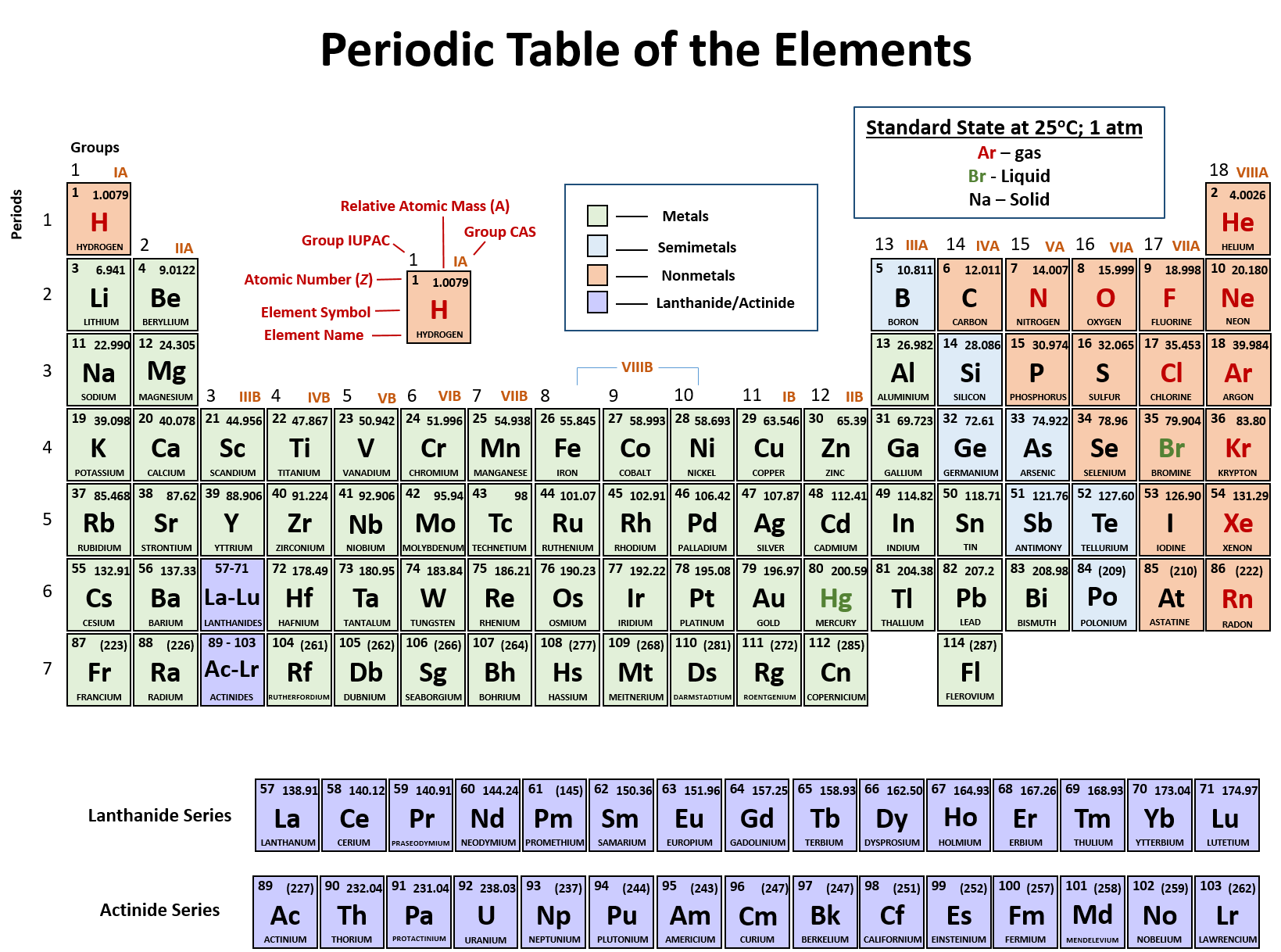
These elements are called metalloids or semi-metals. There are also elements that display properties of both metals and non-metals. The non-metals are on the right side of the separating line.The metals are on the left side of the separating line.In the image below, you can see the location of metals and non-metals on the periodic table. Elements can be divided into two broad categories: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
